Evaporation vs Boiling: The Differences Every Student Must Know
Evaporation vs boiling are two ways liquids turn into gas, yet they work very differently. Understanding the difference helps you explain everyday events, from wet clothes drying to a pot of water bubbling on the stove.
At Selftution.com, science topics like this are broken down in a clear, student-friendly way so learning feels effortless.
What Is Evaporation vs Boiling? A Quick Overview
When scientists talk about phase changes, they mean the process by which matter shifts from one state to another. For example, matter exists in four main states: solid, liquid, gas, and plasma. Both evaporation and boiling convert a liquid into a gas, yet they do so in very different ways.
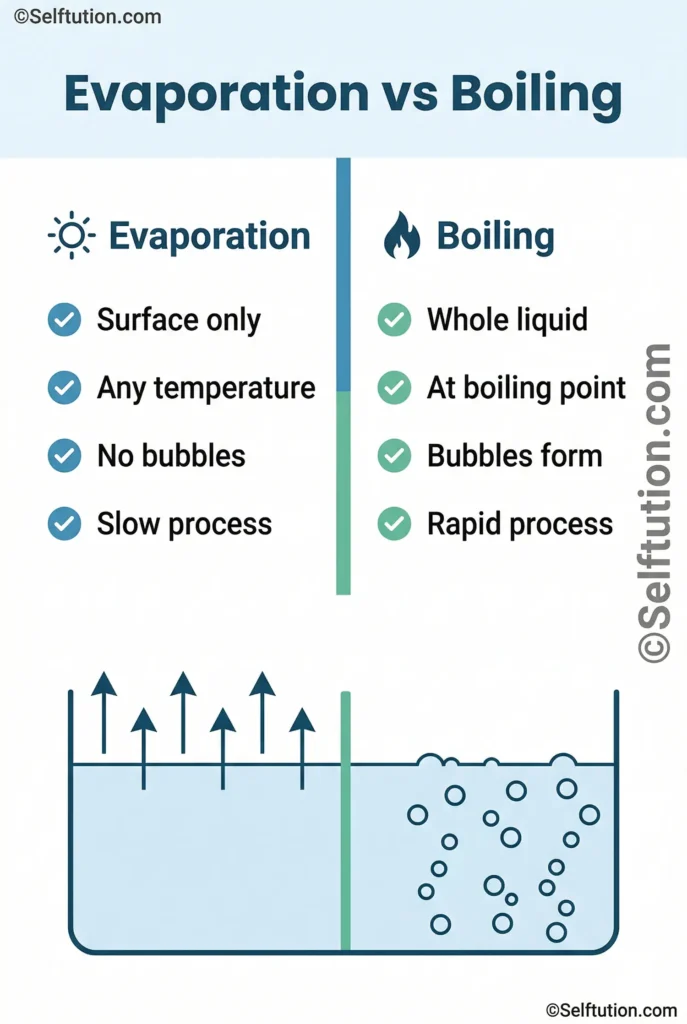
Evaporation is a surface process. It takes place only at the top layer of a liquid, at any temperature above absolute zero. Boiling, however, happens throughout the entire liquid at a specific temperature called the boiling point. Therefore, they are related but not the same.
How Does Evaporation Actually Happen?
Evaporation occurs because liquid molecules are always moving. Some molecules near the surface gain enough heat energy to escape into the air as vapour. This process happens continuously, even in a glass of cold water left on a table.
Several factors speed up evaporation. First, a higher temperature means more molecules have the energy to escape. Second, a larger surface area exposes more molecules to the air. Third, wind or airflow carries vapour away, allowing fresh liquid molecules to evaporate. Additionally, lower humidity in the surrounding air makes it easier for vapour to enter the atmosphere.
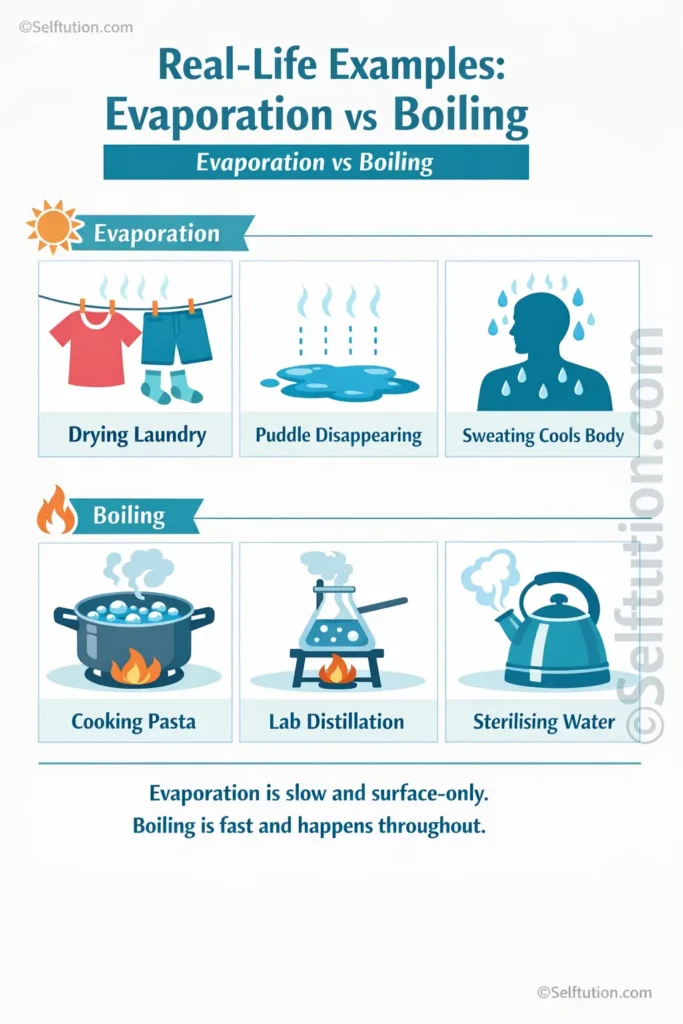
This is why wet clothes dry faster on a warm, windy day than on a cold, still one. Furthermore, this is the same mechanism that drives the water cycle, where oceans and lakes slowly release water vapour into the atmosphere.
Why Boiling Is Different From Evaporation
Boiling is a bulk process. When you heat water on a stove, you supply thermal energy to the entire liquid, not just the surface. As the temperature rises and reaches the boiling point (100°C for water at sea level), vapour bubbles form throughout the liquid and rise to the surface. Consequently, you see the familiar rolling, bubbling action.
The boiling point is not fixed for all conditions. For example, at higher altitudes where air pressure is lower, water boils below 100°C. This is why cooking in the mountains takes longer. Conversely, in a pressure cooker, higher pressure raises the boiling point above 100°C, cooking food faster.
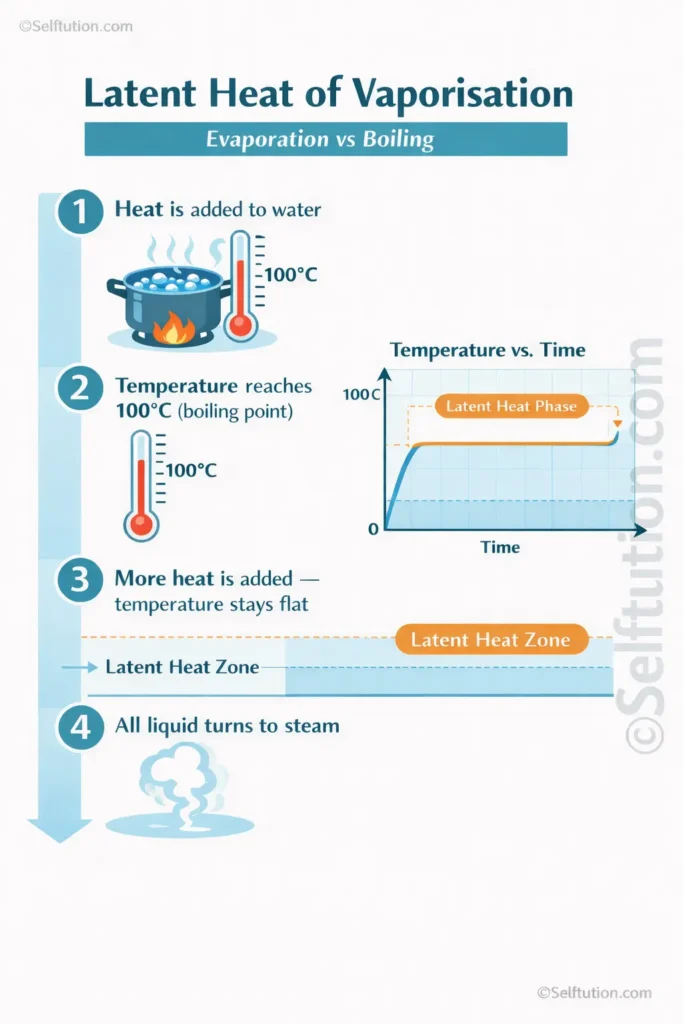
Boiling requires a specific amount of energy called the latent heat of vaporisation. This is the energy needed to change the state of a liquid to a gas without changing its temperature. As a result, water stays at 100°C while it boils, even though heat is still being added.
The Role of Latent Heat in Phase Change
Latent heat is a fascinating concept in science. When water boils, the added energy does not increase the temperature. Instead, it breaks the bonds holding liquid molecules together. Therefore, the temperature stays constant at the boiling point until all the liquid has turned to gas. Exothermic and endothermic reactions follow similar energy principles.
In contrast, evaporation is a cooling process. When fast-moving molecules escape from a liquid’s surface, they take energy with them. This is why you feel cool after swimming as the water evaporates from your skin. Additionally, this principle explains how sweat cools your body on a hot day.
Latent heat of vaporisation
Evaporation vs Boiling: Side-by-Side Comparison
Key differences at a glance:
- Temperature: Evaporation occurs at any temperature; boiling occurs only at the boiling point.
- Location: Evaporation happens at the surface; boiling occurs throughout the liquid.
- Speed: Boiling is a rapid process; evaporation is slow and gradual.
- Bubbles: Boiling produces bubbles inside the liquid; evaporation does not.
- Energy needed: Boiling requires continuous, intense heat; evaporation can occur with ambient energy.
- Everyday example: Drying laundry (evaporation) vs. cooking pasta (boiling).
Furthermore, understanding these differences helps explain many real-world phenomena, from how physical and chemical changes work to why elements and compounds behave differently when heated.
Real-Life Examples That Make the Concepts Click
Evaporation surrounds you every day. After rain, puddles disappear even when the temperature is well below 100°C. Perfume and hand sanitiser evaporate quickly because their molecules are lighter and escape the surface easily. Similarly, the surface of the sea loses enormous amounts of water through evaporation, feeding clouds and rainfall.
Boiling also appears frequently in daily life. Sterilising water kills bacteria reliably because it requires reaching the boiling point. Additionally, industrial processes use boiling to separate substances. For example, in distillation, a mixture is boiled and the vapour is collected and cooled to produce a purified liquid. This is related to the study of pure substances in chemistry and how homogeneous and heterogeneous mixtures behave.
Moreover, both processes are important in understanding the importance of chemistry in everyday life. From cooking and weather to medicine and manufacturing, evaporation and boiling are at work constantly.
Quick Tips to Remember for Your Exam
Use these memory hooks:
- “E” for Escape: Evaporation = surface molecules. Escape slowly, at any temperature.
- “B” for Bulk: Boiling = Bulk heating to the Boiling point, with Bubbles.
- Latent heat rule: Temperature stays flat during boiling because energy goes into breaking bonds, not raising heat.
- Cooling connection: Evaporation cools the surface it leaves behind.
These distinctions are also tested in topics like measurement of temperature and energy transformation, so mastering them now will help you across multiple chapters.
Frequently Asked Questions
1. What is the main difference between evaporation and boiling?
Evaporation happens only at the surface of a liquid and at any temperature. Boiling happens throughout the entire liquid only when it reaches the boiling point, producing bubbles throughout.
2. Can evaporation happen below the boiling point?
Yes. Evaporation can happen at any temperature above absolute zero. For example, a puddle evaporates at room temperature, which is far below 100°C, the boiling point of water.
3. Why does boiling produce bubbles but evaporation does not?
During boiling, vapour forms inside the liquid and rises as bubbles. During evaporation, only surface molecules escape, so there are no bubbles. The bubbles in boiling are a clear sign that vaporisation is happening throughout the liquid.
4. Does evaporation cool the liquid?
Yes. When high-energy molecules escape during evaporation, the remaining liquid loses energy and becomes cooler. This is why sweating cools your body and why a wet cloth feels cold on your forehead.
5. How does air pressure affect boiling?
Lower air pressure lowers the boiling point of a liquid. At high altitudes, water boils below 100°C. Higher pressure raises the boiling point. This is how a pressure cooker works, allowing water to get hotter before it boils.
For further reading, visit the Britannica article on Evaporation