WHAT ARE MOLECULES?
Molecules are the smallest particles of any substance, which have all properties of that particular substance. If we break the molecule further than properties of that substance are lost. A molecule is formed when two or more atoms of the same or different types of elements combine in a fixed ratio. There are 118 different kinds of atoms. Most of these atoms do not like to stay alone. Therefore, they combine with other atoms to form bigger size particle, molecule.

Topics Covered:
- Definition of a molecule
- Difference between atoms and molecules
- Types of molecules
- Importance of the study of molecules
- Types of bonds
Definition of a Molecule
Molecule is the smallest particle of any substance, which is responsible for all physical and chemical properties of that particular substance. It is formed when two or more atoms of the same or different types of elements combine in a fixed ratio.
Let us understand by example.

Sugar Crystals
We all know sugar taste sweet. If we break a crystal of sugar into two pieces and taste them one by one, we will notice that both of them still taste the same i.e. sweet. However, if somehow you break this crystal billion of times up to the smallest particle. You will observe that this smallest particle still tastes sweet. But, if you break this particle further up to the atomic level, then atoms will not be sweet anymore as they have their own set of properties. This smallest particle of sugar, which still tastes sweet, is called a molecule of sugar.
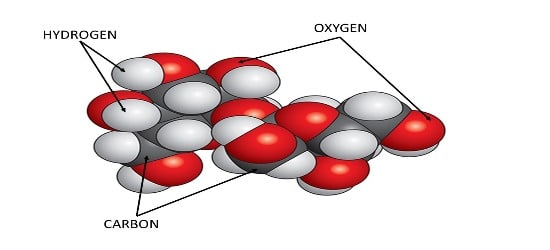
A molecule of Sugar is the smallest particle of sugar crystal. It consists of three kinds of atoms – hydrogen, oxygen & carbon
As most of the atoms do not like to stay alone, thus it is difficult to find a single free atom in nature, except that of inert elements.
A molecule may consist of –
- same kind of two or more atoms, or
- different kinds of two or more atoms
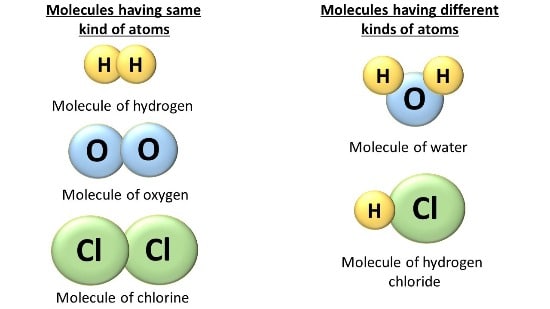
Difference between Atoms & Molecules
Atoms are the basic building block of all substances around us. The different kinds of atoms combine in a fixed ratio to form in numerous molecules and substances.
The important difference between atoms and molecules are as below:
- Atoms are basic building blocks.
- There are only 118 different kinds of atoms.
- Molecules are bigger size particles consisting of two or more atoms of the same or different kind.
- As atoms combine in varying ratios thus, practically uncountable numbers of molecules can be formed.
Type of molecules
Molecules are of three types – monoatomic, diatomic, and polyatomic
- Monoatomic molecules are those which consist of a single atom. For example – Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn).
- Diatomic molecules are those which consist of two atoms of the same or different elements. For example – Hydrogen gas (H2), Oxygen gas (O2), Chlorine (Cl2), Carbon monoxide (CO), Hydrochloric acid (HCL), etc.
- Polyatomic molecules are those which consist of more than two atoms of the same or different elements. For example – Ozone (O3), Phosphorous (P4), Sulphur (S8), Water or Hydrogen monoxide (H2O), Carbon dioxide (CO2), Sodium bicarbonate (NaHCO3), etc. This list can never be exhaustive as still there are innumerous molecules that mankind needs to discover.

Examples of different types of molecules – Monoatomic, Diatomic, and Polyatomic
Importance of the Study of Molecules
The study of molecules is important because the properties of a substance or material depend upon the molecules it consists of. Molecules together make substance, just as bricks make a wall. As the strength and color of the wall will depend upon the kind of bricks used to build it, similar properties of substance will depend upon the kind of molecules used to make it.
Let us take an example, which will clarify things further. When two atoms of hydrogen (H) join, it from a molecule of hydrogen gas (H2). However, when two atoms of hydrogen (H) and one atom of oxygen (O) join, it makes a molecule of water (H2O), which is liquid. Thus, both substances formed had a different set of properties –
- H2 (hydrogen gas) is a gas under normal conditions.
- Hydrogen gas is flammable and when ignited it burns with a pop sound.
- H2O (water) is liquid under normal conditions.
- Another way round we use water to extinguish or control fire.
Type of Bonds
Atoms in a molecule are held together with two types of bonds – ionic bond, and covalent bond.
Atoms combine using the ionic bond when one of the atoms participating in molecule formation releases electrons in the outermost shell and hand it over to the other atoms. Due to this the atom which loses electrons become positively charged and the other which took electrons becomes negatively charged. As particles bearing opposite charges attract each other, thus atoms stay together due to the formation of an ionic bond among them.
Covalent bonds are formed when atoms do not release or take electrons, but instead of that share electrons among them.
















It’s going to be end of mine day, however before end I am reading this wonderful piece of writing
to increase my knowledge.
Excellent post. Ι was checking constantly this blog and Ӏ’m inspired!
Ⅴery helpful іnformation specially tһe
closing ѕection 🙂 I tɑke care of such іnformation а lⲟt.
I used to be seeking tthis сertain information for a vеry lengthy tіme.
Ꭲhank yօu and gooⅾ luck.
I think Lithium does not exist as mono atomic gas molecule; kindly confirm
Yes, you are correct. Like other metals, lithium exists as a solid at room temperature.