Interconversion of States of Matter With Examples
Matter occurs in four states – solid, liquid, gas, and plasma. When matter changes its state from one form to the other it is known as the interconversion of states of matter. But before we proceed with the interconversion of the state of matter, let us learn, what is matter and what is the difference between these four states of matter.
When you look around, you see many things like plants, animals, land, water, rocks, and different objects like desks, chairs, books, pens, bags, shoes, houses, cars, etc. But have you wondered what makes up plants and animals? What makes up land and water? What makes up a desk and chair? The answer to all of these questions is matter. Everything in the Universe, from the tiniest particle of dust to the biggest star, is made up of matter.
So, the matter is not only what we can see or touch, but it also includes the air we breathe in, the perfume we smell, etc. Therefore, we can say that all physical substances which we can see, touch, smell, or feel around us are matter.
DEFINITION OF MATTER
The definition of matter for kids –
The matter is anything which occupies space and has a mass.
In the post “What is an atom?” we learned that the atom is the main building block of all things around us. However, it will be interesting for you to know that even atoms and other subatomic particles are made up of matter.

Experiment to demonstrate that air is a form of matter which occupies space and has mass
CAN MATTER BE CREATED?
The Universe consists of two things – matter and energy. Most scientists believe that matter and energy are the same things and each can convert into the other. During the Big Bang at the time of the creation of the Universe, a great amount of energy evolved. Then just after a few seconds, some bundles of energy turned into tiny particles of matter. These tiny particles combined to form atoms that made up the Universe we live in today. All the matter created during the Big Bang still exists. It can be as huge as a planet or a star, or as small an atom – or even as tiny as the subatomic particles. This matter exists in four states – solid, liquid, gas, and plasma, which undergo interconversion depending upon prevailing physical conditions. The interconversion of states of matter occurs without a change in its chemical composition.
FOUR STATES OF MATTERS
Since ancient times, people believed that matter exists only in three states or forms – solid, liquid, and gas. However, this changed in the 1920s with the discovery of the fourth state of matter – plasma. Plasma is a very uncommon state of matter. It exists only at incredibly high temperatures, in nuclear reactors, or inside stars.
Sometimes, the same matter can exist in three different states. For example, water is a liquid normally; if we fill it in an ice tray and keep it in the freezer for some time, it becomes solid (ice). The same water, when boiled for some time, changes into a gas (steam). Although, each state of matter has its unique features and properties. But the atoms and molecules in the matter do not change for each different state. When a substance changes from one state to another, without a change in its chemical composition, it is called the interconversion of states of matter.
We can explain the different states of matter based on the arrangement of particles or molecules in it. Molecule of any kind of matter always attracts each other with a force – an intermolecular force. The force of attraction between two molecules increases if the space between them decreases and vice-versa. The space between the molecules is called intermolecular space.
The molecules of matter are always in random motion because they possess kinetic energy. The kinetic energy increases with temperature and vice-versa. Due to random motion, these molecules sometimes come closer and sometimes move apart.
SOLIDS

In solids, the molecules are very close to each other. There is a strong force of attraction between the molecules and the space between them is very small (almost negligible). The molecules are, therefore, not free to move. They merely vibrate at their positions. This makes solids hard and difficult to compress, giving them a fixed shape and size.
LIQUIDS

In the case of liquids, the molecules are not as close to each other as in solids. They also do not attract each other as strongly as the molecules of solids. Thus the spaces between them are larger and molecules can move about more freely. This makes a liquid flow and takes the shape of the container into which it is poured. Thus, liquids have fixed volumes but no definite shape of their own.
GASES

Molecules in Gases are far away from each other
In the case of gas, the molecules hardly attract each other. They lie far apart from each other and the space between them is very large. The force of attraction is so weak that the molecules have great freedom to move. As a result, gases have neither a fixed shape nor a fixed volume. They fill up the space available to them. Due to the huge space between molecules of gas, we can easily compress the gas.
PLASMA

Cloud of Plasma
Plasma unlike the other three states of matter is not made of atoms or molecules. It is a state of matter, which is somewhat like gas but its properties are significantly different from that of gas. In plasma, atoms lose electrons and become positively charged, while the electrons move freely. It is an electrically neutral gas with both positively and negatively charged particles (ions) moving freely in the same space. Plasma is a very uncommon state of matter. It exists only at incredibly high temperatures, in nuclear reactors, or inside stars. As per scientists, 99% of the matter in the Universe is plasma.
COMPARISON BETWEEN PROPERTIES OF SOLIDS, LIQUID, GASES, AND PLASMA
The property-wise comparison between four states of matter i.e. solid, liquid, gas, and plasma are tabulated below:

Comparison between four states of matter
It is important to note that, four states of matter – solid, liquid, gas, and plasma, undergo interconversion depending upon prevailing physical conditions.
INTERCONVERSION OF STATES OF MATTER
When matter changes its state from one form to the other it is known as the interconversion of states of matter.
The matter or substance can change state, from solid to liquid, or from liquid to gas, and vice-versa. In everyday life, we come across substances that change from one state to another. For example, water is a liquid under normal conditions, but when cooled it turns into ice. This happens because during cooling we remove kinetic energy from molecules of water in the form of heat. This increases the force of attraction between the molecules and brings them closer to form ice, a solid. Contrary to this, when we heat water, it starts boiling and turns into steam. During boiling, we provide kinetic energy to the molecules of water in the form of heat. This weakens the intermolecular force of attraction between molecules and they fly further apart from each other in the form of steam, a gas.
Similarly, steam on cooling turns into liquid water, and so does ice when kept at room temperature. But the properties of water remain the same in all three states.
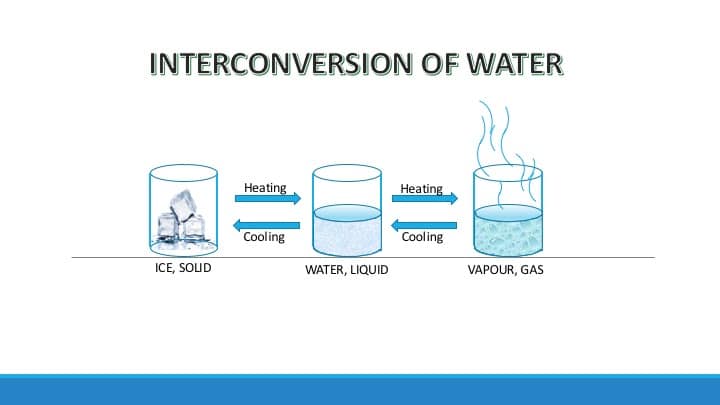
The picture depicts the interconversion of states of matter with an example of water. Here water, a form of matter undergoes interconversion from solid to liquid state and from liquid to gaseous state on heating. Similarly, on cooling vapor changes to a liquid and on further cooling to a solid.
Video showing interconversion of states of matter. Courtesy Amrita Vishwa Vidyapeetham University
SOME TERMS RELATED TO INTERCONVERSION OF STATE OF MATTER
- Melting or Fusion: It is a process by which a substance changes from a solid to a liquid state on heating.
- Melting Point: It is the temperature at which a pure substance changes from a solid to a liquid state.
- Vapourization or Evaporation: It is a process by which a substance changes from liquid to gaseous state on heating at any temperature.
- Boiling: It is a process by which a substance changes from liquid to a gaseous state on heating at a fixed temperature.
- Boiling point: It is the temperature at which a pure substance changes from a liquid to a gaseous state.
- Condensation or liquefaction: It is a process by which a substance changes from a gas to a liquid state on cooling at a fixed temperature.
- Solidification or freezing: It is a process by which a substance changes from liquid to a solid state on cooling















Good day! Do you use Twitter? I’d like to follow you
if that would be okay. I’m definitely enjoying your blog and look
forward to new posts.