BASIC STRUCTURE OF AN ATOM FOR KIDS
This post explains the basic structure of an atom concerning subatomic particles (electrons, protons, and neutrons) present in it.
TOPICS COVERED:

Representation of an atom
1.0 INTRODUCTION – THE BASIC STRUCTURE OF AN ATOM
Atoms are the basic building block of matter. They are the smallest particles, which has all the features and properties of that particular element. Although atoms are the smallest block it is not like that that we can’t break them. Yes, you read it right. Atoms are not the smallest particles. We can break an atom into subatomic particles – electrons, protons, and neutrons. However, it is important to note that if we break an atom, then all features of that particular element are lost. Thus, before unraveling, the basic structure of an atom, let us learn who discovered the subatomic particles.
2.0 DISCOVERY OF SUBATOMIC PARTICLES
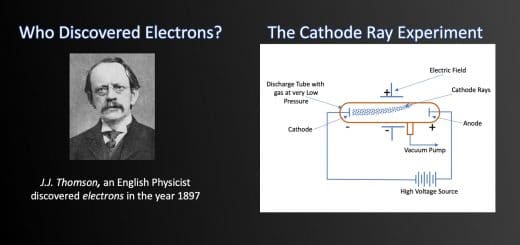
For a long period in history, scientists were of opinion that atoms could not be broken further into smaller particles. But things changed after the discovery made by a scientist named J.J. Thomson in the year 1897. In his quest to ascertain the basic structure of an atom, he discovered that atoms are divisible and contain a negatively charged subatomic particle – ‘electron’. To know more about J.J. Thomson’s discovery, click here.
Soon, after J.J. Thomson’s discovery, the race began among other scientists to uncover the basic structure of an atom.
- Goldstein in the year 1898, concluded that – since all atoms are electrically neutral, thus, there must be positively charged particles present in them. To know more about Goldstein’s experiment, click here.
- Rutherford discovered the nucleus in 1911 and protons in 1917. He further stated that these positively charged particles join together to form a ‘nucleus’. This nucleus is present at the center of an atom and all electrons revolve around it. To know more, click here.
- James Chadwick in the year 1932, studied the atomic model and discovered particles that have no charge but have mass almost equal to that of the protons. He named them – ‘neutron’. To know more, click here.
Back to the introduction – Structure of an atom
2.0 BASIC STRUCTURE OF AN ATOM
According to the modern standard model, the basic structure of an atom is as follows:
- An atom consists of the subatomic particles called electrons, protons and neutrons.
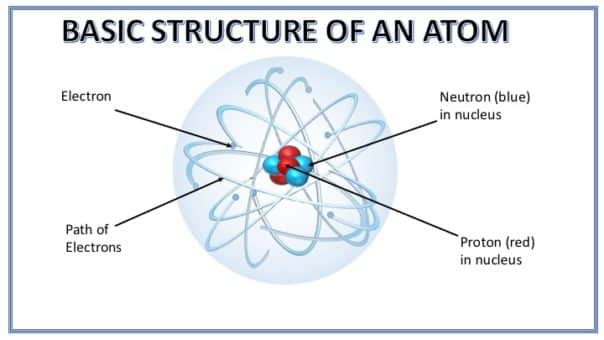
- There are two parts of an atom – the nucleus and the orbits or the shells.
- The nucleus is the central part of the atom. This consists of protons and neutrons. Each proton has an electrical charge, almost like a tiny battery, but not both positive and negative – only positive. Thus, the nucleus bears a positive charge on it. Neutrons are of the same size as protons but they have no electrical charge. As each proton has a positive charge on them, thus they tend to repel each other. Neutrons help in keeping protons together. The entire mass of an atom lies in its nucleus as electrons have negligible mass. The mass of a proton and neutron is approximately 1837 times of that of electrons.
- Electrons are much smaller than protons and neutrons. They are not present in the nucleus. They whizz around the nucleus in space, in imaginary layers known as orbits or shells. Each orbit is associated with a fixed amount of energy. Therefore, these circular orbits are also known as energy levels or energy shells. Electrons in the outer shells have more energy than those in the inner shells. Each electron also has an electric charge, which is negative – opposite to the charge of a proton.
- Usually, an atom has the same number of protons and electrons. So, the positives and negatives are equal, which means the whole atom has no electrical charge.
3.0 PROPERTIES OF ELECTRONS, PROTONS, AND NEUTRONS
3.1 PROPERTIES OF ELECTRONS
- Electrons are an integral part of all atoms.
- An Electron has a definite mass and it carries a definite electric charge.
- The mass of an electron is 1/1837 of the mass of a hydrogen atom (9.108 x 10-28 g).
- It has one unit negative charge which is equal to 1.602 x 10-19 Coulombs.
- An electron is denoted by the symbol -1e0. The superscript ‘0’ represents its mass and the subscript ‘-1’ represents it’s one unit negative electrical charge.
3.2 PROPERTIES OF PROTONS
- The mass of a proton is nearly equal to the mass of an atom of hydrogen, i.e. 1.672 x 10-24 g.
- The positive charge with a proton is equal to the negative charge on an electron, i.e. 1.602 x 10-19 Coulombs.
- A proton is denoted as +1p1, where the superscript ‘1’ represents 1 amu (atomic mass unit) mass and the subscript ‘+1’ represents it’s one unit positive charge.
3.3 PROPERTIES OF NEUTRONS
- The mass of a neutron is slightly more than that of a proton, i.e. 1.676 x 10-24 g compared to 1.672 x 10-24 g.
- Electrically, a neutron is neutral, which means it has no charge.
Back to the introduction – Structure of an atom
4.0 ATOMS OF DIFFERENT ELEMENTS
There are 118 different kinds of atoms. These atoms make 118 kinds of basic substances, elements. In every atom of every chemical element, each kind of sub-atomic particle is the same. So the electrons in an atom of iron are the same as the electron in an atom of oxygen. The protons in carbon atoms are the same as the protons in hydrogen atoms. And the neutrons in an atom of carbon are the same as that of atoms of iron. So, important to understand here is that, what makes these chemical elements different is how many subatomic particles they have in them.
The basic structure of an atom of elements differs from each other in respect to the number of protons, neutrons, and electrons it contains. For example, the smallest atom of hydrogen contains only one proton, one electron, and no neutron. Whereas the largest known atom of Ununoctium (also known as Oganesson) posses 118 protons, 118 electrons and approximately 176 neutrons in it.
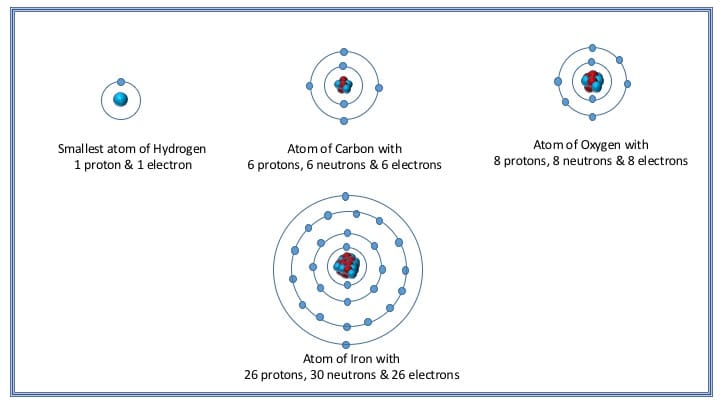
The basic structure of hydrogen, carbon, oxygen & Iron atom
5.0 STRUCTURE STABILITY OF AN ATOM
We know that there exists a force of attraction between particles with opposite electrical charges. Thus, there is a force of attraction between the electrons and the protons present within an atom. Electrons revolve around the nucleus at a fixed distance from it. To avoid fall into the nucleus, electrons revolve at a very high speed. Electrons revolve around the nucleus at speed approximately 3/4th of the speed of light. Electrons with low speed or energy revolve closer to the nucleus in comparison to those with high energy levels. However, it is expected that electrons being lighter, charged and in constant motion, would gradually lose energy and come closer to the nucleus and eventually fall into it, thus resulting in the structural collapse of the atom. But this does not happen.
According to Bohr’s theory, the electrons revolved in fixed orbits or shells around the nucleus at a very high speed, with each orbit associated with a fixed amount of energy. The electrons present in these shells, neither lose nor gain energy until some external force is applied to it. Thus, they maintain their position. As a result, the inward force exerted by the nucleus is counterbalanced by the outward force of the moving electrons, thus preventing the electrons from falling into the nucleus and making the atom structurally stable.
Back to the introduction – Structure of an atom
6.0 ELECTRON SHELLS
Wonderful video showing the basic structure of an atom. Courtesy St. Mary’s Physics Online
To understand the basic structure of an atom, good knowledge of electron shells is very important. The existence of electron Shells was first brought out by Charles Brakla. He stated there are 7 numbers of electron shells and labeled them with letters K, L, M, N, O, P and Q. They are also labeled as 1, 2, 3, 4, 5, 6, and 7 going from innermost shell to outwards. The closest shell to the nucleus is ‘1-shell’ or ‘K-shell‘, next comes ‘2-shell‘ or ‘L-shell‘ and so on. The farthest shell from the nucleus is ‘7-shell’ or ‘Q-shell’.
Each type of shell can contain only a fixed number of electrons. To determine this number we use formula 2n². Here ‘n’ stands for the numeric label of the shell. From the formula, the maximum number of electrons in shell K is 2, L is 8, M is 16, N is 32, O is 50, P is 72 and Q is 98.
Although, the formula gives the maximum in principle, in fact, that maximum is only achieved for the first four shells (K, L, M, N). No known element has more than 32 electrons in any one shell. This is because of the presence of sub-shells.
To know more about electron shells, click here
7.0 FUNDAMENTAL OR ELEMENTARY PARTICLES
For a long time in history, atoms were believed as fundamental or elementary particles of the Universe. However, this belief changed after unraveling the basic structure of an atom and discovery of subatomic particles – electrons, protons, and neutrons. Now with advances in science, we know that there are many other subatomic particles besides electrons, protons, and neutrons. They include muons, gluons, gravitons and dozens of others. And even particles protons and neutrons are not the smallest of all. They are made of even tinier pieces – quarks. There are six kinds of quarks, with the odd names of up, down, strange, charmed, bottom and down quark. Quarks, along with a group of particles called leptons, which includes electrons, are probably the smallest pieces of matter. They might be fundamental or elementary particles.
To know more about fundamental or elementary particles, click here
What we learned above was very basic about the structure of an atom. Keep following for related posts.
















9 Responses
[…] The basic structure of an atom […]
[…] Basic Structure of an atom for kids […]
[…] or to ancient Greek philosopher Democritus (470 – 400 BC). But, only a few people know that atoms were first discovered by the great Indian philosopher Maharishi Kanad in 600 BC. He named them […]
[…] a long period in history, scientists were of opinion that atoms could not be broken further. But things changed after the discovery made by an English scientist […]
[…] The basic structure of atoms […]
[…] The basic structure of an atom […]
[…] a long period in history, scientists were of opinion that atoms could not be broken further. But things changed after the discovery made by an English scientist […]
[…] discovered negatively charged electrons, Eugen Goldstein, a German concluded that since all atoms are electrically neutral, thus there must be positively charged particles present in them. He […]
[…] […]